For as long as I can remember, I've been impressed by Gaboon Vipers (Bitis gabonica). These western African behemoths can reach 5 3/4 feet in length and over 14.5 inches in girth, and weigh up to 25 pounds with an empty stomach. They are the heaviest vipers and possess the longest fangs, up to one and a half inches in length! Furthermore, their geometric dorsal pattern, as intricate as it is beautiful, is ideally suited to camouflaging them against the leafy forest floor, where they lie in wait for their endothermic prey: birds, rodents, rabbits, monkeys, small antelope, porcupines.
| A Gaboon Viper, beautifully camouflaged |
Like many vipers, Gaboon Vipers are ambush predators, a lifestyle to which they are supremely adapted. Long folding fangs and deadly venom allow them to kill their prey while keeping a safe distance from it. A very low metabolism permits them to wait in one spot for weeks, until the perfect opportunity presents itself. They spend between three-quarters and 95% of their time just sitting quietly, sometimes for up to three months at a time. Every so often, a viper, particularly a male during the breeding season of March through May, will embark on a long-distance movement of one quarter to two thirds of a mile, sometimes in a single day.1 The preferred habitat of Gaboon Vipers is a mosaic of forest, thicket, and grassland, although they will sometimes enter sugarcane fields and rural gardens. As with most snakes, life as a Gaboon Viper is probably pretty dull.
 |
| The impressive fangs of a Gaboon Viper |
 |
| You can see why |
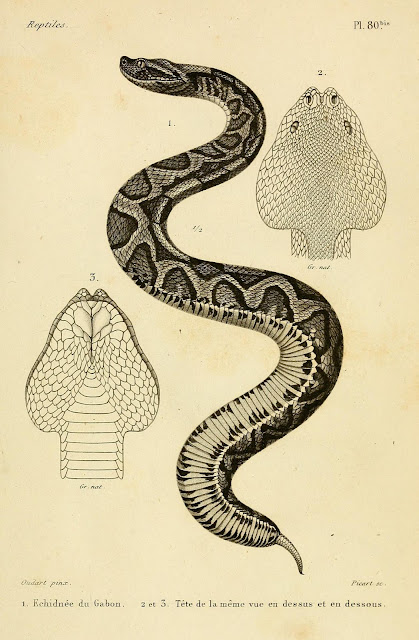 |
| Gaboon Viper plate from Duméril, Bibron, & Duméril's Erpétologie Générale; unfortunately, this is one of the only plates not in color |
 |
| East African Gaboon Viper (B. g. gabonica) |
 |
| West African Gaboon Viper (B. g. rhinoceros) |
I learned something new about these vipers recently. It seems that, among other heavy-bodied snakes, they have evolved the ability to retain their feces for incredibly long periods of time - months to years, after which time 5-20% of the body weight of a single snake may be feces. While this would kill a human, retained fecal material may be functioning as metabolically inert ballast in these species, which require a stationary inertial base for striking. Available data suggest that enhanced uptake of water and nutrients can also be achieved in snakes retaining feces - the poisonous urates (read: pee) are excreted more frequently. Amazing.
1 One exception is that these snakes always move following shedding, which occurs about twice a year, perhaps to distance themselves from potential predators attracted by the sloughed material or to avoid external parasites in the old skin that could reattach to the snake.↩
2 Snake biologists in Africa face challenges unfamiliar to we North Americans: "In several instances, I had to abort tracking efforts due to B. gabonica locations in close proximity to potentially dangerous game; namely [Water Buffalo, Rhinoceros, Elephant, Hippopotamus, and Crocodile]", writes Jonathan Warner in his dissertation.↩
ACKNOWLEDGMENTS
Thanks to Tim Vickers, Wolfgang Wuster, Ivica, Markus Oulehla, and Jonathan Warner for their photos.
REFERENCES
Lillywhite HB, de Delva P, Noonan BP (2002) Patterns of gut passage time and chronic retention of fecal mass in viperid snakes. In: Schuett GW, Höggren M, Douglas ME, Greene HW (eds) Biology of the Vipers. Eagle Mountain Publishers, Eagle Mountain, UT, pp 497-506
Linn I, Perrin M, Bodbijl T (2006) Movements and home range of the gaboon adder, Bitis gabonica gabonica, in Zululand, South Africa. Afr Zool 41:252-265
Luiselli L (2006) Site occupancy and density of sympatric Gaboon viper (Bitis gabonica) and nose-horned viper (Bitis nasicornis). J Trop Ecol 22:555-564
Luiselli L (2006) Site occupancy and density of sympatric Gaboon viper (Bitis gabonica) and nose-horned viper (Bitis nasicornis). J Trop Ecol 22:555-564
Marsh NA, Whaler BC (1984) The Gaboon Viper (Bitis gabonica): Its biology, venom components and toxinology. Toxicon 22:669-694
Warner JK (2009) Conservation Biology of the Gaboon Adder (Bitis gabonica) in South Africa. PhD dissertation, School of Animal, Plant, and Environmental Sciences, University of the Witwatersrand, Johannesburg, South Africa.
 |
| This Gaboon Viper quilt was made for me by my mother on my 21st birthday. The geometric pattern lends itself perfectly to quilting. |
Life is Short, but Snakes are Long by Andrew M. Durso is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.








